- Home
- Technical Information
- Analysis Technology
TECHNICAL INFOMATION
Analysis Technology
Equipment Owned by the R&D Center
Fabricating prototype materials
Aluminum alloys that have been adjusted to the desired composition are melted and casted in various types of molds to fabricate prototype materials. Heat treatment is performed if necessary.
-
[Melting and casting]

Melting furnaces for testing (electric furnaces)
Melting amount: Approximately 1 to 50 kg
-
[Heat treatment]

Furnaces for solution and aging treatments
Hot-air-circulation electrically-heated oven (550˚C maximum)
Evaluation of material properties
Test specimens are obtained from prototype materials in accordance with various tests to evaluate the mechanical properties, microstructures and casting properties of aluminum alloys.
-
[Mechanical properties]

Tensile testing machine
Room temperature, high temperature
(10-t maximum load for both) -
[Mechanical properties]

Hardness testing machine
Brinell (HB)
Rockwell (HRB)
Micro-Vickers
(HV0.025 to 1) -
[Mechanical properties]

Impact testing machine
5 kg, 10 kg
-
[Mechanical properties]

Abrasion tester
Ogoshi method
-
[Microstructure observation]

Stereoscopic microscope
Magnification from 0.67 to 4.5 times
-
[Microstructure observation]

Optical microscope
Magnification from 50 to 1000 times
-
[Microstructure observation]

Scanning electron microscope (EDS attached)
Magnification from 30 to 300,000 times, detectable elements Be to U
-
[Casting properties]

Fluidity testing machine
MIT method
-
[Casting properties]

Shrinkage characterization
Tatur mold method
-
[Casting properties]

Cooling curve
Apparatus for measuring cooling curve
Ingot/product quality evaluation
Quantitative analysis of component elements in aluminum alloys is carried out with an optical emission spectroscopic analysis method and various wet chemical analysis methods. In addition, the amount of gas and inclusion in molten aluminum alloy is measured.
-
[Molten metal quality]

Hydrogen analyzer
Inert gas fusion-thermal conductivity method
-
[Molten metal quality]

Inclusion evaluation
K-mold method
-
[Elemental analysis]

Optical emission spectrophotometer
11 elements regulated by JIS, and P, Na, Ca, Sr, etc.
-
[Elemental analysis]

Atomic absorption spectrometer
Cu, Mg, Zn, Mn, and many more
-
[Elemental analysis]

Ultraviolet and visible absorption spectrophotometer
Fe, Ti, P, B
Inclusions analysis
Inclusions vary in type and shape, and can be introduced via many routes. In general, they are oxides, carbides, and coarse intermetallic compounds. An example of analysis of oxide inclusions is shown.

SEM image
(magnification: 1000 times)
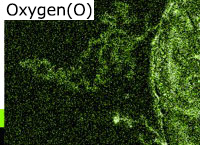
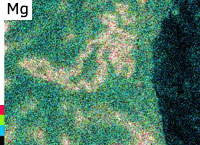
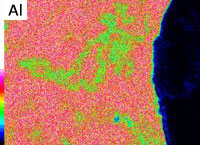
EDS elemental analysis
Chemical Analysis
Wet chemical analysis
The sample is dissolved with an acid solution, etc., and the quantity of the target element is measured with a weighing method, etc. The amounts of the trace elements in alloys are quantified with high precision based on stoichiometry.
We produce nominal standard samples by setting component values in wet chemical analysis, and use them for our alloy ingot quality control.

Hydrogen analysis
Hydrogen analysis
Hydrogen accounts for the majority of the gas that dissolves into aluminum. Compared to the liquid phase, the solubility of hydrogen in the solid phase is extremely low. If a large amount of hydrogen gas is contained in the molten metal, gas pores will form in the ingots, adversely affecting their quality. Therefore, it is extremely important for the quality control for ingots to correctly evaluate the amount of hydrogen gas in the molten metal.
In addition to the qualitative and/or semi-quantitative analysis methods used at the production site, there are laboratory analysis methods for quantitatively evaluating the amount of hydrogen gas in aluminum with high precision.
In an inert gas carrier extraction method, which is a laboratory analysis method, a solid sample is melted in an inert gas stream to extract hydrogen gas. The carrier gas's thermal conductivity is different when it contains hydrogen gas. The change in thermal conductivity is converted into the amount of hydrogen gas. This method is widely used because it is easy to perform and can be used to carry out analysis in less time.